Phase Change and Latent Heat
- There is chemical potential energy in the weak van der Waals attractions as well as in the strong ionic and covalent bonds.
- Because the van der Waals attraction is so much weaker, there is much less chemical potential possible here, but it is a major factor in the energy of phase changes.
- As you can see below, the chemical potential energy of water is less than that of water vapor (or steam) which helps to explain why steam burns can be so severe. Not only is the steam hot, but it tends to condense into a liquid when it touches something. When forming a liquid the chemical potential energy that is released when the van der Waals attractions form can add substantial heat energy to the process of heat transfer from the water molecules to whatever they are condensing on.
- See a diagram of the energy below:
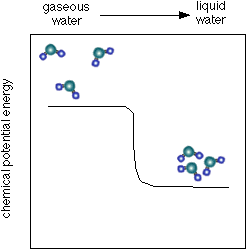
- A process that converts chemical potential energy into heat energy is called exothermic.
- A process that converts heat energy to chemical potential energy is called endothermic.
- Whenever a change of state occurs energy is either absorbed or released
- An exothermic process releases heat (converting some of its chemical potential energy to heat energy).
- An endothermic process absorbs heat (converting some of the heat energy into chemical potential energy.)
- Below is a table of common phase changes and their associated energy
Process Name of phase change Energy flow solid -> liquid 
liquid -> solid 
liquid -> gas 
gas -> liquid 
solid -> gas 
gas -> solid 
- In endothermic phase changes the heat energy is used to overcome the weak intermolecular attractions.
- In exothermic phase changes some chemical potential energy is converted to heat energy when the weak intermolecular attractions form.
- Latent Heat is the term describing the energy you get from exothermic phase changes. There are two phase changes where this heat energy is released:
- Condensation: When gas condenses to liquid the quantity of energy converted from chemical to heat is called the Heat of Vaporization or Δ Hv.
- Freezing: When a liquid freezes into a solid the quantity of energy converted from chemical to heat is called the Heat of Fusion or Δ Hf.